Step therapy criteria
Step Therapy Criteria 2013 4 Tier Standard Last Updated: 02/11/2013 ANGIOTENSION RECEPTOR BLOCKERS Products Affected Criteria Step 1: First line therapy should be losartan or losartan/hctz. Step 2: Second line therapy should be Benicar or Benicar HCT. Step 3: Once losartan or losartan/hctz and Benicar or Benicar HCT have been tried, patients can receive therapy with Diovan o
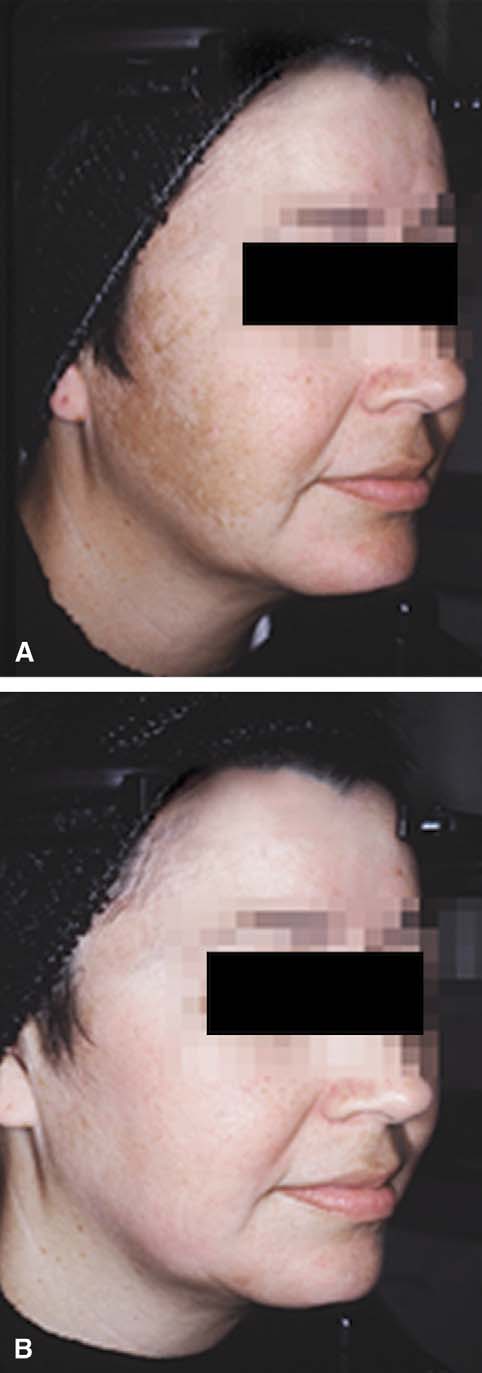 decreasing cellular metabolism. Tretinoin has beenfound to abrogate the epidermal atrophy that canoccur with topical corticosteroids. Various modifica-tions to the original formula have subsequently beenstudied.
decreasing cellular metabolism. Tretinoin has beenfound to abrogate the epidermal atrophy that canoccur with topical corticosteroids. Various modifica-tions to the original formula have subsequently beenstudied.