msszinfo.hu
A Reergin tabletta kétféle amonisavat (glutaminsavat és aszparaginsavat) tartalmaz. Anaerob edzéseknél elősegíti a bomlástermékek (elsősorban a tejsav) átalakítását és kiürítését. A szellemi munkára kedvezőtlenül hat. 2. Inozin tabletta Az inozin emeli a sejtek ATP (adenozin-trifoszfát) szintjét, amely a rövid ideig tart
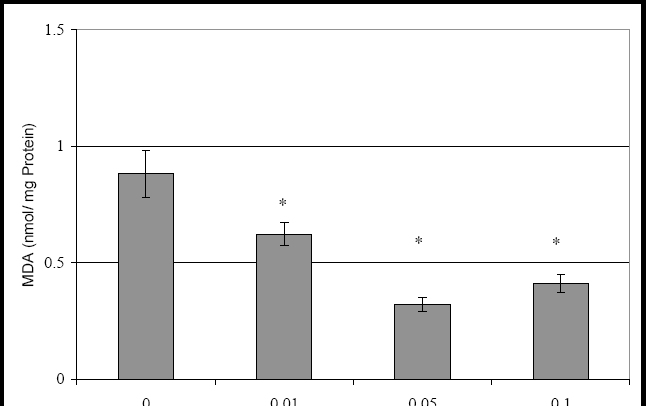

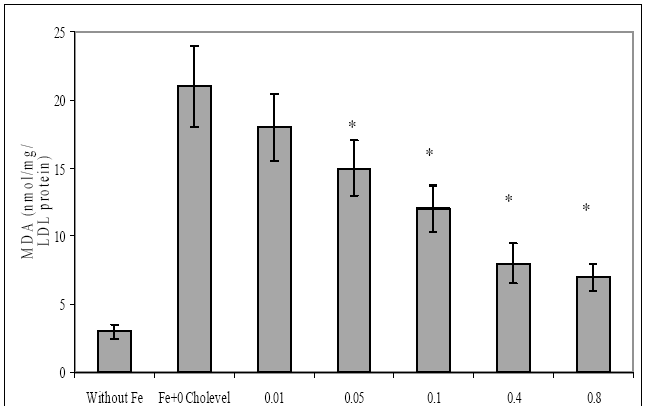
 88 The Open Complementary Medicine Journal, 2009, Volume 1
88 The Open Complementary Medicine Journal, 2009, Volume 1